Promising Preliminary Results in Optic Nerve Recovery Study of NurExone’s first product ExoPTEN for Glaucoma
Minimally Invasive treatment with ExoPTEN showed functional restoration of damaged eyes to healthy levels in animals
TORONTO and HAIFA, Israel, July 17, 2024 (GLOBE NEWSWIRE) -- NurExone Biologic Inc. (TSXV: NRX), (OTCQB: NRXBF), (Germany: J90) (the “Company” or “NurExone”), a pioneering biopharmaceutical company, is pleased to announce the preliminary results from a small-scale controlled study exploring the use of its flagship nanodrug, ExoPTEN, for optic nerve recovery in a rat model at Sheba Medical Center. This study marks a second clinical indication being investigated for ExoPTEN.
The study was initiated by Professor Michael Belkin, following the success of ExoPTEN in nerve regeneration in the spinal cord indication in preclinical models. An Optic Nerve Crush (“ONC”) model was used to simulate conditions like glaucoma, where the optic nerve is crushed, resulting in impaired vision.
Glaucoma is a common eye condition, particularly in older adults typically caused by optic nerve compression and pressure in the eye. The prevalence of glaucoma in the Western world is generally estimated to be around 2-3% in people aged 40 and older. The risk increases with age, and the prevalence can be higher in populations over 60. Estimated Number of People Affected in the United States alone, is over 3 million people, with many more cases likely undiagnosed.
The study carried out under Prof. Ygal Rotenstreich and Dr. Ifat Sher of Sheba Medical Center explored the therapeutic effects of ExoPTEN on retinal function after ONC compared to healthy baseline levels, an untreated ONC control and ONC treated with naïve exosomes. Importantly ExoPTEN was administered minimally-invasively using suprachoroidal injection in a delivery system invented by Prof. Rotenstreich.
As expected, the post-ONC control eyes exhibited a marked decline in retinal functionality, as evidenced by the lack of a peak (Fig. A – red graph). Experimental treatments with ExoPTEN (“ONC+PTEN"), showed promising results, with treated eyes exhibiting a peak similar to the healthy eye in the same animal indicating recovery of retinal response following optical nerve compression (Fig. B – green graph). The naïve exosome-treated rats (“ONC+EXO”) showed a lower peak and increased latency indicating a weaker response (Fig. C – brown graph). The results presented are from just 18 days following the ONC damage. These treatment findings suggest potential pathways for recovery of optic nerve function and overall healthy vision.
Dr. Ifat Sher and Prof Ygal Rotenstreich of Sheba Medical Center commented: "While these results are preliminary, they form a solid foundation for further research. Our next steps include more extensive studies to validate these findings and explore their potential application for humans."
Dr. Lior Shaltiel, CEO of NurExone, added: "We are excited by these preliminary findings and commend the team at Sheba for this work, which is an important step in our mission to develop regenerative therapies. These early studies suggest potential for ExoPTEN in the US$3.4 billion glaucoma market and will allow us to help those affected by retinal degenerative conditions."
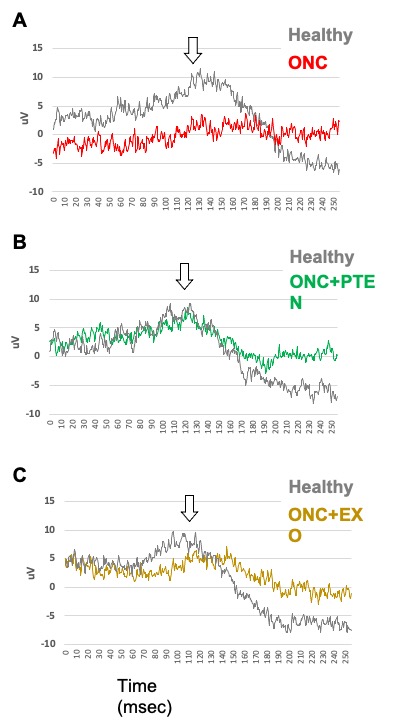
Figure 1 – Positive impact of specific treatments on retinal health
Graphs A-C show Electroretinogram (ERG) measurements of dark-adapted (scotopic) threshold retinal response (STR, in microvolts, V) at -36 dB of three representative rats. In each rat, one eye was left intact as a healthy control (“Healthy”, gray). Rat A had ONC in one eye (red) with no treatment, which resulted in a flat, near-zero retinal response. Rat B had ONC in one eye and was treated with ExoPTEN (green, ONC+PTEN), resulting in a retinal response similar to the healthy intact contralateral eye. Rat C had ONC in one eye and was treated with naïve exosomes (brown, ONC+EXO), resulting in a recordable but delayed and smaller retinal response compared to the healthy control contralateral eye. The results are following the minimally-invasive administration of two treatment cycles (one post-operation and the other in the subsequent week), with a volume of 20μL per eye in the treated and the control rats (naïve exosomes).
About NurExone Biologic Inc.
NurExone Biologic Inc. is a TSXV listed pharmaceutical Company that is developing a platform for biologically-guided exosome-based therapies to be delivered, non-invasively, to patients who have suffered Central Nervous System injuries. The Company’s first product, ExoPTEN for acute spinal cord injury, was proven to recover motor function in 75% of laboratory rats when administered intranasally. ExoPTEN has been granted Orphan Drug Designation by the FDA. The NurExone platform technology is expected to offer novel solutions to drug companies interested in noninvasive targeted drug delivery for other indications.
For additional information, please visit www.nurexone.com or follow NurExone on LinkedIn, Twitter, Facebook, or YouTube.
For more information, please contact:
Dr. Lior Shaltiel
Chief Executive Officer and Director
Phone: +972-52-4803034
Email: info@nurexone.com
Thesis Capital Inc.
Investment Relation - Canada
Phone: +1 905-347-5569
Email: IR@nurexone.com
Dr. Eva Reuter
Investment Relation - Germany
Phone: +49-69-1532-5857
Email: e.reuter@dr-reuter.eu
FORWARD-LOOKING STATEMENTS
This press release contains certain “forward-looking statements” that reflect the Company’s current expectations and projections about its future results. Wherever possible, words such as “may”, “will”, “should”, “could”, “expect”, “plan”, “intend”, “anticipate”, “believe”, “estimate”, “predict” or “potential” or the negative or other variations of these words, or similar words or phrases, have been used to identify these forward-looking statements. Forward-looking statements in this press release include, but are not limited to, statements the scope and benefits of the stated study, including the proposed subsequent studies; the statement that the NurExone platform technology is expected to offer novel solutions to drug companies interested in noninvasive targeted drug delivery for other indications, including recovery of optic nerve function and overall visual health; the website and its benefits; and the Company’s ongoing commitment to using its ExoTherapy platform and nanodrug ExoPTEN to treat acute spinal cord injuries.
These statements reflect management’s current beliefs and are based on information currently available to management as at the date hereof. In developing the forward-looking statements in this press release, we have applied several material assumptions, including the Company’s ability to carry out the scope and realize upon the benefits of the stated study; the Company’s ability to realize upon the stated potential for its nanodrug ExoPTEN to treat acute spinal cord injuries; the Company’s ability to realize the NurExone platform technology to offer novel solutions to drug companies interested in noninvasive targeted drug delivery for other indications.; the new website being more clear, intuitive, and easy to follow for collaborators, pharmaceutical companies, and other stakeholders; and the Company’s ability to maintain its ongoing commitment to using its ExoTherapy platform to advance the field of regenerative medicine.
Forward-looking statements involve significant risk, uncertainties and assumptions. Many factors could cause actual results, performance or achievements to differ materially from the results discussed or implied in the forward-looking statements. These risks and uncertainties include, but are not limited to, risks related to the Company’s inability to carry out the scope and realize upon the benefits of the stated study; lack of revenues to date; government regulation; market acceptance for its products; rapid technological change; dependence on key personnel; the fact that its nanodrug ExoPTEN is still under development; the fact that the effects of the nanodrug ExoPTEN are still uncertain; the potential inability to obtain or maintain regulatory approval of ExoPTEN; the introduction of competing drugs that are safer, more effective or less expensive than, or otherwise superior to, the drug product candidates of the Company; potential inability to obtain adequate financing; dependence on the Company’s strategic partners; the protection of the Company’s intellectual property; the potential inability to obtain or maintain intellectual property protection for the ExoTherapy platform and ExoPTEN; risks that the Company’s intellectual property and technology won’t have the intended impact on the Company and/or its business; the website not being more clear, intuitive, or easy to follow for collaborators, pharmaceutical companies, and other stakeholders; and the risks discussed under the heading “Risk Factors” on pages 29 to 36 of the Company’s Annual Information Form dated March 30, 2023, a copy of which is available under the Company’s SEDAR+ profile at www.sedarplus.ca. These factors should be considered carefully, and readers should not place undue reliance on the forward-looking statements. Although the forward-looking statements contained in this press release are based upon what management believes to be reasonable assumptions, the Company cannot assure readers that actual results will be consistent with these forward-looking statements. These forward-looking statements are made as of the date of this press release, and the Company assumes no obligation to update or revise them to reflect new events or circumstances, except as required by law.
Neither TSXV nor its Regulation Services Provider (as that term is defined in the policies of the TSXV) accepts responsibility for the adequacy or accuracy of this release.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/ed7191aa-533d-40b4-b8ba-1aa6ebe2c6f6